JEE Advance - Chemistry (2023 - Paper 2 Online)
2
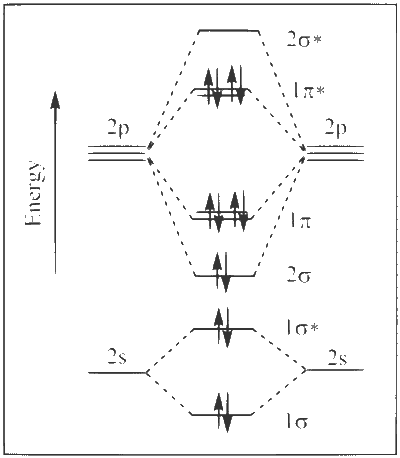
Consider the following statements related to colloids.
(I) Lyophobic colloids are not formed by simple mixing of dispersed phase and dispersion medium.
(II) For emulsions, both the dispersed phase and the dispersion medium are liquid.
(III) Micelles are produced by dissolving a surfactant in any solvent at any temperature.
(IV) Tyndall effect can be observed from a colloidal solution with dispersed phase having the same refractive index as that of the dispersion medium.
The option with the correct set of statements is :
(I) Lyophobic colloids are not formed by simple mixing of dispersed phase and dispersion medium.
(II) For emulsions, both the dispersed phase and the dispersion medium are liquid.
(III) Micelles are produced by dissolving a surfactant in any solvent at any temperature.
(IV) Tyndall effect can be observed from a colloidal solution with dispersed phase having the same refractive index as that of the dispersion medium.
The option with the correct set of statements is :
Answer
(A)
(I) and (II)
6
Atoms of metals $\mathrm{x}, \mathrm{y}$, and $\mathrm{z}$ form face-centred cubic (fcc) unit cell of edge length $\mathrm{L}_{\mathrm{x}}$, body-centred cubic (bcc) unit cell of edge length $\mathrm{L}_y$, and simple cubic unit cell of edge length $\mathrm{L}_z$, respectively.
If $\mathrm{r}_{\mathrm{z}}=\frac{\sqrt{3}}{2} r_{\mathrm{y}} ; \mathrm{r}_{\mathrm{y}}=\frac{8}{\sqrt{3}} \mathrm{r}_{\mathrm{x}} ; M_{\mathrm{z}}=\frac{3}{2} M_{\mathrm{y}}$ and $M_{\mathrm{z}}=3 M_{\mathrm{x}}$, then the correct statement(s) is(are) :
[Given: $M_x, M_y$, and $M_z$ are molar masses of metals $x, y$, and $z$, respectively.
$\mathrm{r}_{\mathrm{x}}, \mathrm{r}_{\mathrm{y}}$, and $\mathrm{r}_{\mathrm{z}}$ are atomic radii of metals $\mathrm{x}, \mathrm{y}$, and $\mathrm{z}$, respectively.]
If $\mathrm{r}_{\mathrm{z}}=\frac{\sqrt{3}}{2} r_{\mathrm{y}} ; \mathrm{r}_{\mathrm{y}}=\frac{8}{\sqrt{3}} \mathrm{r}_{\mathrm{x}} ; M_{\mathrm{z}}=\frac{3}{2} M_{\mathrm{y}}$ and $M_{\mathrm{z}}=3 M_{\mathrm{x}}$, then the correct statement(s) is(are) :
[Given: $M_x, M_y$, and $M_z$ are molar masses of metals $x, y$, and $z$, respectively.
$\mathrm{r}_{\mathrm{x}}, \mathrm{r}_{\mathrm{y}}$, and $\mathrm{r}_{\mathrm{z}}$ are atomic radii of metals $\mathrm{x}, \mathrm{y}$, and $\mathrm{z}$, respectively.]
Answer
A
B
D
8
$\mathrm{H}_2 \mathrm{S}$ (5 moles) reacts completely with acidified aqueous potassium permanganate solution. In this reaction, the number of moles of water produced is $\mathbf{x}$, and the number of moles of electrons involved is $\mathbf{y}$. The value of $(\mathbf{x}+\mathbf{y})$ is ________.
Answer
18
9
Among $\left[\mathrm{I}_3\right]^{+},\left[\mathrm{SiO}_4\right]^{4-}, \mathrm{SO}_2 \mathrm{Cl}_2, \mathrm{XeF}_2, \mathrm{SF}_4, \mathrm{ClF}_3, \mathrm{Ni}(\mathrm{CO})_4, \mathrm{XeO}_2 \mathrm{~F}_2,\left[\mathrm{PtCl}_4\right]^{2-}, \mathrm{XeF}_4$, and $\mathrm{SOCl}_2$, the total number of species having $s p^3$ hybridised central atom is ________.
Answer
5
10
Consider the following molecules: $\mathrm{Br}_3 \mathrm{O}_8, \mathrm{~F}_2 \mathrm{O}, \mathrm{H}_2 \mathrm{S}_4 \mathrm{O}_6, \mathrm{H}_2 \mathrm{S}_5 \mathrm{O}_6$, and $\mathrm{C}_3 \mathrm{O}_2$. Count the number of atoms existing in their zero oxidation state in each molecule.
Their sum is _______.
Their sum is _______.
Answer
6
11
For $\mathrm{He}^{+}$, a transition takes place from the orbit of radius $105.8 \mathrm{pm}$ to the orbit of radius $26.45 \mathrm{pm}$. The wavelength (in nm) of the emitted photon during the transition is _______.
[Use :
Bohr radius, $\mathrm{a}=52.9 \mathrm{pm}$
Rydberg constant, $R_{\mathrm{H}}=2.2 \times 10^{-18} \mathrm{~J}$
Planck's constant, $\mathrm{h}=6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$
Speed of light, $\mathrm{c}=3 \times 10^8 \mathrm{~m} \mathrm{~s}^{-1}$ ]
[Use :
Bohr radius, $\mathrm{a}=52.9 \mathrm{pm}$
Rydberg constant, $R_{\mathrm{H}}=2.2 \times 10^{-18} \mathrm{~J}$
Planck's constant, $\mathrm{h}=6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$
Speed of light, $\mathrm{c}=3 \times 10^8 \mathrm{~m} \mathrm{~s}^{-1}$ ]
Answer
30
12
$50 \mathrm{~mL}$ of 0.2 molal urea solution (density $=1.012 \mathrm{~g} \mathrm{~mL}^{-1}$ at $300 \mathrm{~K}$ ) is mixed with $250 \mathrm{~mL}$ of a solution containing $0.06 \mathrm{~g}$ of urea. Both the solutions were prepared in the same solvent. The osmotic pressure (in Torr) of the resulting solution at $300 \mathrm{~K}$ is _______.
[Use: Molar mass of urea $=60 \mathrm{~g} \mathrm{~mol}^{-1}$; gas constant, $\mathrm{R}=62$ L Torr $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$;
Assume, $\Delta_{\text {mix }} \mathrm{H}=0, \Delta_{\text {mix }} \mathrm{V}=0$ ]
[Use: Molar mass of urea $=60 \mathrm{~g} \mathrm{~mol}^{-1}$; gas constant, $\mathrm{R}=62$ L Torr $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$;
Assume, $\Delta_{\text {mix }} \mathrm{H}=0, \Delta_{\text {mix }} \mathrm{V}=0$ ]
Answer
682
13
The reaction of 4-methyloct-1-ene $(\mathbf{P}, 2.52 \mathrm{~g})$ with $\mathrm{HBr}$ in the presence of $\left(\mathrm{C}_6 \mathrm{H}_5 \mathrm{CO}\right)_2 \mathrm{O}_2$ gives two isomeric bromides in a $9: 1$ ratio, with a combined yield of $50 \%$. Of these, the entire amount of the primary alkyl bromide was reacted with an appropriate amount of diethylamine followed by treatment with aq. $\mathrm{K}_2 \mathrm{CO}_3$ to give a non-ionic product $\mathbf{S}$ in $100 \%$ yield.
The mass (in mg) of $\mathbf{S}$ obtained is ________.
[Use molar mass (in $\mathrm{g} \mathrm{mol}^{-1}$ ) : $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{Br}=80$ ]
The mass (in mg) of $\mathbf{S}$ obtained is ________.
[Use molar mass (in $\mathrm{g} \mathrm{mol}^{-1}$ ) : $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{Br}=80$ ]
Answer
1791
16
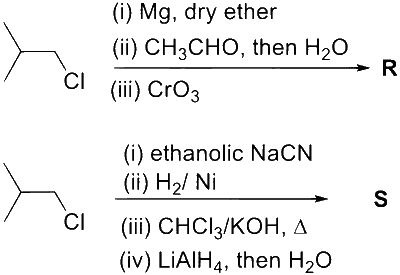
The number of heteroatoms present in one molecule of $\mathbf{R}$ is _________.
[Use : Molar mass (in $\left.\mathrm{g} \mathrm{mol}^{-1}\right)$ : $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{O}=16, \mathrm{Br}=80, \mathrm{Cl}=35.5$
Atoms other than $\mathrm{C}$ and $\mathrm{H}$ are considered as heteroatoms]
[Use : Molar mass (in $\left.\mathrm{g} \mathrm{mol}^{-1}\right)$ : $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{O}=16, \mathrm{Br}=80, \mathrm{Cl}=35.5$
Atoms other than $\mathrm{C}$ and $\mathrm{H}$ are considered as heteroatoms]
Answer
9
17
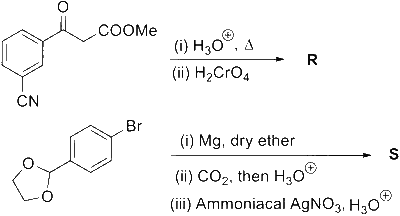
The total number of carbon atoms and heteroatoms present in one molecule of $\mathbf{S}$ is _________.
[Use : Molar mass (in $\mathrm{g} \mathrm{mol}^{-1}$ ): $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{O}=16, \mathrm{Br}=80, \mathrm{Cl}=35.5$
Atoms other than $\mathrm{C}$ and $\mathrm{H}$ are considered as heteroatoms]
[Use : Molar mass (in $\mathrm{g} \mathrm{mol}^{-1}$ ): $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{O}=16, \mathrm{Br}=80, \mathrm{Cl}=35.5$
Atoms other than $\mathrm{C}$ and $\mathrm{H}$ are considered as heteroatoms]
Answer
51