JAMB - Chemistry (2025 - No. 98)
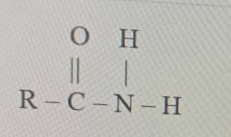
The above structure is
Explanation
The structure RCONH₂ contains a carbonyl group (C=O) bonded directly to a nitrogen atom (NH₂). This specific functional group, CONH₂, is characteristic of an amide. In the general naming convention for this class of organic compounds, they are known as alkanamides.
Alkylamine: Amines have a general formula of RNH₂ (primary amine) without the carbonyl group (C=O) adjacent to the nitrogen.
Alkanone: Alkanones, also known as ketones, have a carbonyl group (C=O) bonded to two other carbon atoms (RCOR'), and do not contain a nitrogen atom.
Amino acid: Amino acids are organic compounds that contain both an amino group (–NH₂) and a carboxylic acid group (–COOH) attached to the same central carbon atom, along with a hydrogen atom and a variable side chain (R group). The structure RCONH₂ lacks a carboxyl group.
The correct option is C. alkanamide.
Comments (0)


