JAMB - Chemistry (2025 - No. 68)
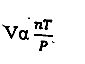
The expression above represents
Avogadro's law
Boyle's law
General gas law
Charles' law
Explanation
The general gas law equation, often called the Ideal Gas Law, is PV = nRT, describing the relationship between a gas's Pressure (P), Volume (V), amount in mole(n), Ideal gas constant(R), and Temperature (T), relating all four key properties in one formula. A related form, the combined law, for a fixed mass of gas is (P₁V₁)/T₁ = (P₂V₂)/T₂, used to find changes in one variable when others change. However, the equation given in the question will give PV = nRT, on rearrangement.
Comments (0)


