JAMB - Chemistry (2025 - No. 52)
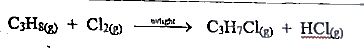
The reaction above is
neutralization
substitution
polymerization
oxidation
Explanation
Propane, C\(_3\)H\(_8\) reacting with chlorine ((Cl\(_2\)) in sunlight is a free radical substitution reaction, also known as photochemical chlorination, where sunlight (UV light) provides energy to break the \(Cl-Cl\) bond, allowing chlorine atoms to replace hydrogen atoms on the propane molecule, forming chloropropanes and hydrogen chloride (HCl).
Comments (0)


