JAMB - Chemistry (2025 - No. 21)
Which of the following pairs of elements will exhibit diagonal relationship?
Be and Si
Na and Be
Li and Al
B and Si
Explanation
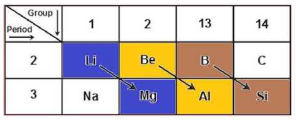
Diagonal relationships in the periodic table occur between certain pairs of elements in the second and third periods that are diagonally adjacent to each other. These pairs exhibit similar chemical and physical properties due to comparable ionic sizes and charge/radius ratios.
The following pairs exhibit diagonal relationship in the periodic table; Li and Mg, Be and Al, B and Si. Hence, the correct answer is B and Si- Option D
Comments (0)


