JAMB - Chemistry (2025 - No. 100)
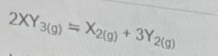
In the equation above, the expression for the equilibrium constant, k\(_c\) is
\(\frac{[2XY_3]}{[X_2][3Y_2]}\)
\(\frac{[X_2][3Y_2]}{2XY_3]}\)
\(\frac{[XY_3]^2}{[X_2][Y_2]}\)
\(\frac{[X_2][Y_2]^3}{[XY_3]^2}\)
Explanation
The equilibrium constant expression, based on the law of mass action, is defined as the ratio of the concentrations of the products raised to their stoichiometric coefficients to the concentrations of the reactants raised to their stoichiometric coefficients.
The correct answer is option D - \(\frac{[X_2][Y_2]^3}{[XY_3]^2}\)
Comments (0)


